
Achieving a Diagnosis
You Can Help Identify DMD With a Creatine Kinase (CK) Test
A CK test is a quick and often inexpensive tool1 that can potentially prevent other unnecessary testing.1
The CK test is the first step in determining whether a neuromuscular condition is causing developmental delays and muscle weakness.2
The CK test measures the amount of CK in the blood. CK is an enzyme that is found in skeletal muscle, cardiac muscle, and the brain.3 It is released from muscle cells into the blood during muscle injury.4 Elevated levels almost always indicate skeletal muscle damage and can be observed as early as at birth.1,5
CK Levels are not checked as part of preventative pediatric care6
Although the normal range of CK levels varies with age and gender, CK levels in DMD are typically elevated 50- to 200-fold above normal levels, as shown in the table below.1
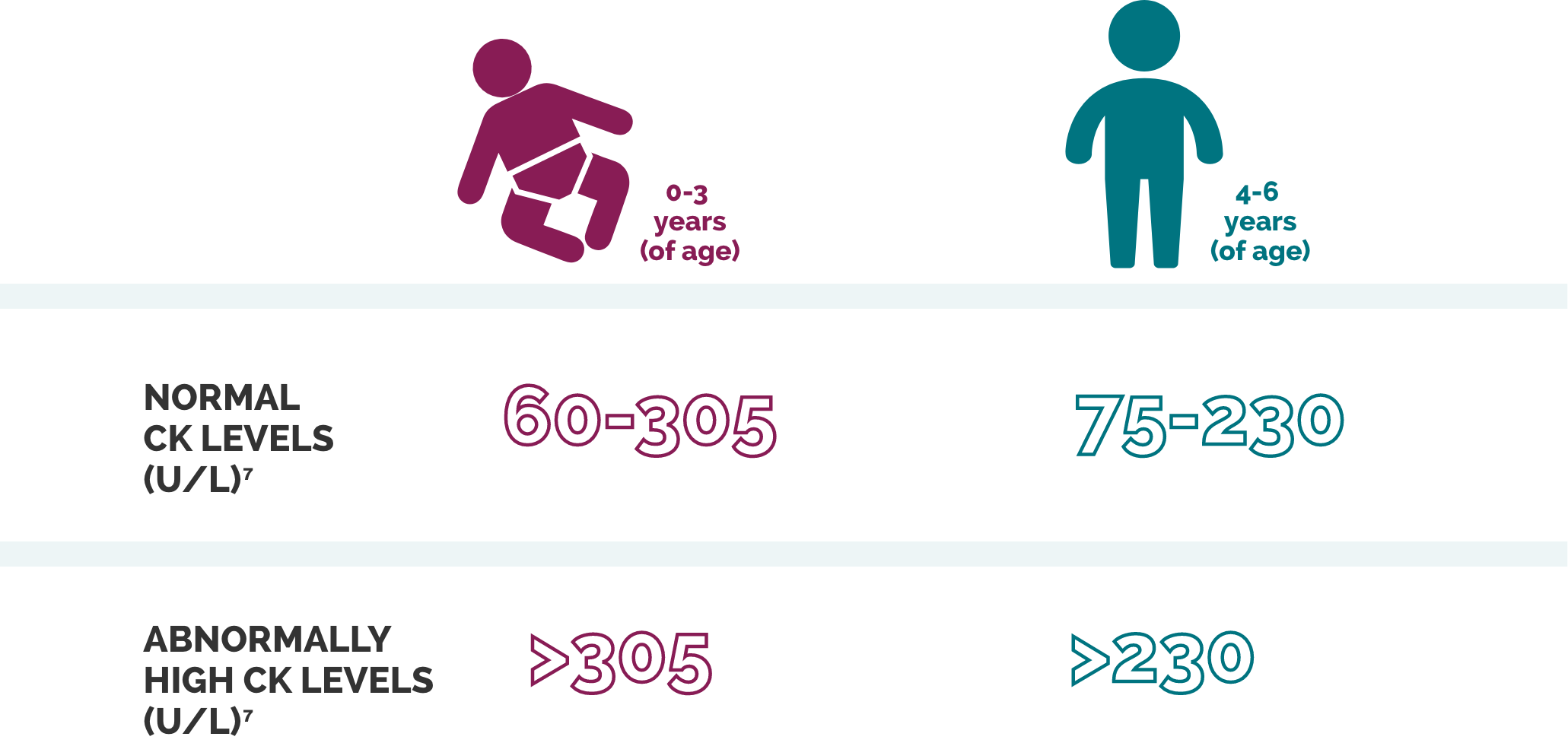
The ICD-10* diagnosis code for muscle weakness is M62.81.8
If CK is elevated, refer to a neuromuscular specialist to confirm a diagnosis.12
*International Classification of Diseases, 10th revision.
Diagnosing DMD with Genetic Tests
If CK is elevated, the neuromuscular specialist will order genetic testing to determine whether DMD is the cause of your patient's symptoms.9 A genetic test can be used to confirm that your patient has a DMD genetic mutation and to identify the specific type of mutation.10 Types of genetic tests that can be used include, but are not limited to, the following:4
- Multiplex ligation-dependent probe amplification (MLPA) or array-based comparative genomic hybridization (aCGH)
These tests can identify large-scale mutations in the DMD gene, which are found in ~80% of patients. For the remaining ~20% of patients, additional genetic testing may be needed. - Next-generation sequencing (NGS)
Identifies both large- and small-scale mutations in the DMD gene. Typically, no further genetic testing is required.
In rare cases, genetic testing may be inconclusive. In these situations, the specialist may recommend a muscle biopsy to detect the presence of dystrophin.10
Genetic Testing Helps Determine Treatment Eligibility
Genetic testing is crucial in DMD because certain therapies are only indicated to treat DMD caused by mutations in specific regions of the DMD gene. Some therapies may also have contraindications against specific mutations, or the location of mutations within the DMD gene.4
Some of these mutations may also be too small to detect using MLPA or aCGH technologies.10 NGS can provide both a diagnosis, and therapeutic amenability information necessary to determine if a patient may be amenable to treatment with current or future DMD therapies.4
Sponsored Genetic Testing Programs May Be Available
Sarepta Therapeutics partners with Parent Project Muscular Dystrophy (PPMD) and Decode Duchenne, a genetic testing program that provides free genetic testing to eligible patients who have been unable to access testing due to financial barriers.



